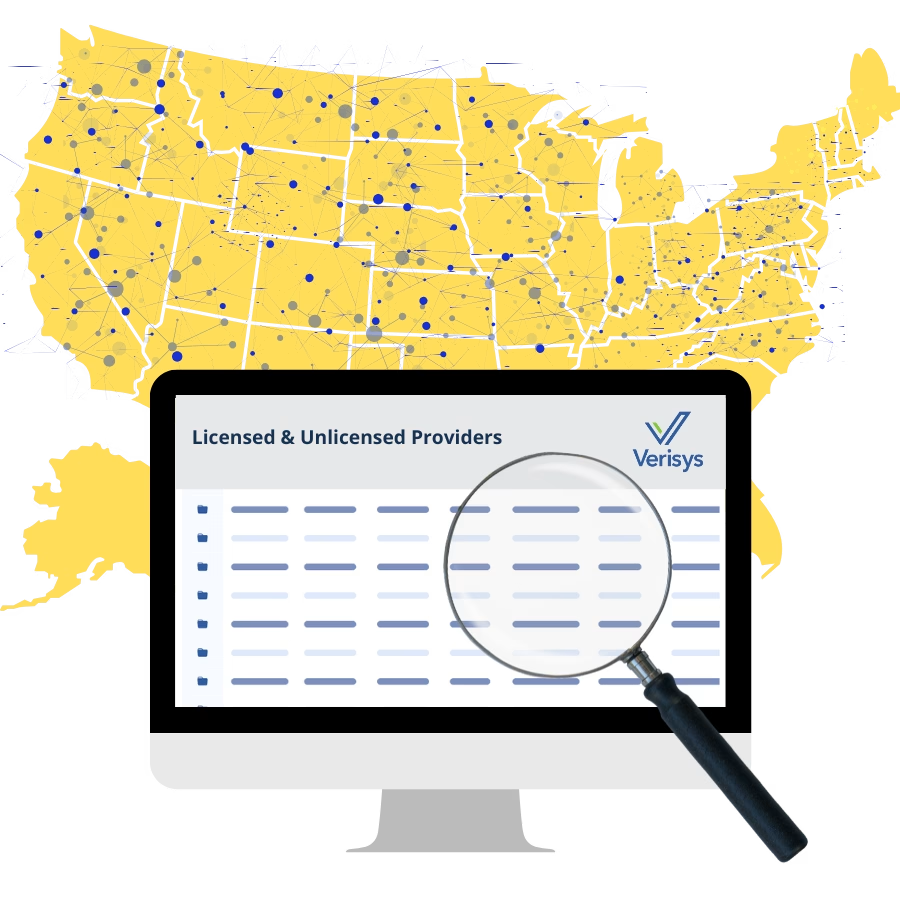
Provider network monitoring requires continuous oversight of every provider in an organization’s network to verify credentials, detect sanctions and exclusions, and confirm...
Verisys Blog
How OIG Monitoring Strengthens Patient Safety and Compliance
OIG monitoring is the recurring process of screening employees, contractors, providers, and vendors against the OIG List of Excluded Individuals and Entities (LEIE) to identify...
Best Provider Network Management Solutions for Multi-State Healthcare Organizations
Managing provider networks across multiple states requires more than maintaining a provider roster. Health plans, managed care organizations, and other healthcare organizations...
Navigating Compliance Challenges for Healthcare Staffing Agencies
Healthcare staffing agencies are under pressure to place qualified providers quickly while maintaining compliance standards that can withstand client, regulatory, and audit...
How Negligent Credentialing Increases Medical Malpractice Exposure
Negligent credentialing exposes healthcare organizations to direct legal liability beyond any individual physician's malpractice. When a facility fails to properly verify and...
Why Provider Eligibility Matters in Workers’ Compensation Claims
Provider eligibility in workers’ compensation is not just a credentialing requirement. It directly determines whether a claim can be paid. When eligibility is not verified...
What Healthcare Organizations Look for in a Compliance Data Provider
Healthcare organizations face many complexities in compliance monitoring: more regulations, more provider types, more data sources, and higher consequences for gaps in...
The Benefits of Continuous Background Checks in Healthcare Staffing
A background check confirms who a provider is at the moment of hire, but it does not confirm who they remain over time. In the healthcare industry, that gap creates measurable...
How Health Plans Can Meet 2026 Network Adequacy Requirements
Medicaid certification for network adequacy is entering a new era of regulation. Beginning in 2026, health plans face stricter federal standards, independent validation...
FACIS vs Other Healthcare Exclusion Databases
For healthcare compliance leaders, exclusion screening is not a routine administrative task; it is one of the most scrutinized elements of regulatory oversight. Civil monetary...